All Features
Dawn Bailey
‘We didn’t get here on our own,” said Brian Dieter, president and CEO of Baldrige Award-recipient Mary Greeley Medical Center (MGMC), speaking at the 32nd Baldrige Quest for Excellence Conference. “We think we are very much better as a result of having learned from [other Baldrige Award recipients…

Rich Tree
Following any tech transfer project, the subsequent startup of the manufacturing line is almost always full of challenges. The goal is to start up as soon as possible once the project is completed but also to achieve steady-state throughput as quickly as possible after the startup begins. This type…

Dirk Dusharme
Like it or not, work-from-home (WFH) is here to stay. This is not just a perk that employers might offer, but a requirement on which many employees, current and future, are demanding. According to several surveys, between 30 and 50 percent of employees surveyed said they would leave their jobs if…

Jay Arthur—The KnowWare Man
There are many control chart rules to detect special causes (i.e., out-of-control conditions). Although most of these rules are clear, the one that seems to befuddle most people is the rule about trends. Is it six points (including the first point), six points (excluding the first point), or seven…

Taran March @ Quality Digest
In regulated industries, every step of the production process must be verified to some sort of guidance or standard. What this comes down to, practically speaking, is an enormous amount of time and effort spent on actions outside the sphere of production. Every day of production seems to create a…

Ravi Anupindi
Inoculating the planet from Covid-19 presents an unprecedented logistical challenge like none we’ve seen before. Mobilizing for a world war may be the closest comparison, but in this case, the enemy is invisible and everywhere.
Some of the vaccines require super-cold storage at virtually all…

Sara Adams
Designing, developing, and getting your medical device approved and onto the market is a huge accomplishment—but it isn’t the end of your responsibilities.
For the European Union (EU) market, the European Commission (EC) requires manufacturers to perform ongoing postmarket surveillance (PMS)…
Dawn Bailey
‘What would be important to you in the care of your daughter?” John Chessare, president and CEO of Baldrige Award-recipient GBMC HealthCare (GBMC), asked a virtual Quest for Excellence conference audience.
“The No. 1 answer is always that people want the best possible health outcome for their…

Boris Babic, Sara Gerke, Theodoros Evgeniou, I. Glenn Cohen
For many of us, our electronic device can be a communications lifeline, entertainment system, and professional networking hub. If trends continue, it may become our health advisor as well.
Direct-to-consumer (DTC) medical apps are a growing segment of the $10 billion market for healthcare…

Zach Winn
This story was originally published by MIT News.
For small healthcare groups like dentist’s offices, one sick staff member can mean a day’s worth of cancelled appointments. Such offices can either continue short-staffed, which could negatively affect patient care, or reschedule appointments,…

Tom Siegfried, Knowable Magazine
This story was originally published by Knowable Magazine.
From the earliest days of their evolution, guts and brains have been the best of friends.
It’s a mutually beneficial relationship. Guts prepare nourishment for delivery to the brain. And brains guide the behaviors needed to fill the gut…

Saligrama Agnihothri
Health-tracking devices and apps are becoming part of everyday life. More than 300,000 mobile phone applications claim to help with managing diverse personal health issues, from monitoring blood glucose levels to conceiving a child.
But so far the potential for health-tracking apps to improve…

Dawn Bailey
The spirit of service—for a small clinic started in 1913 to provide free care to Los Angeles (LA)—lives today in the servant-leader aspirations of 2019 Baldrige Award recipient Adventist Health White Memorial (AHWM), a 353-bed, safety-net hospital.
The community of two million people that AHWM…
Clare Naden
Travel and tourism took a beating during the Covid-19 pandemic, with borders closed, airlines grounded, and many establishments shut for months. Now as the industry attempts to recover in this new context, constantly changing rules and regulations are making it a far-from-simple task. What’s more,…
Adrian Hernandez, C. Michael White
T
he U.S. Food and Drug Administration (FDA) regularly inspects manufacturing facilities to ensure that drugs meet rigorous quality standards. These standards are vital to protect patients from drugs that are incorrectly dosed, contaminated, or ineffective.
But over the past few years, tens of…

Gleb Tsipursky
When the Covid pandemic swept through the country last year, companies rapidly transitioned employees to working from home (WFH). However, this shift led to growing challenges of WFH burnout and Zoom fatigue.
Unfortunately, organizations treat these issues as day-to-day challenges, instead of…

Jon Speer
Demonstrating identification and traceability in all quality system processes is a must for medical device companies to comply with FDA regulations. To satisfy this compliance need, companies will need to connect related processes within their quality system to close the loop between related pre-…

Wade Schroeder
Medical-device usability testing and validation are critical tasks leading up to a medical device’s debut on the market. “Usability” looks at how the user interacts with your device and forms a key component of overall risk management and safety.
If there’s any “spoiler alert” to this article, it’…

Ariane Ollier-Malaterre, Knowable Magazine
This story was originally published by Knowable Magazine.
Working from home—formally known as telework—is here to stay. A 2021 survey of approximately 30,000 Americans concluded that, after the pandemic, 20 percent of all work days may continue to take place at home, vs. just 5 percent before.…

David L. Chandler
This story was originally published by MIT News.
Two MIT professors have proposed a new approach to estimating the risks of exposure to Covid-19 under different indoor settings. The guideline they developed suggests a limit for exposure time, based on the number of people, the size of the space,…

Jon Speer
The medical device technical file is a must-have document for devices to be sold in the European Union (EU) marketplace. The file contains detailed information about your medical device, its design, intended use claims, composition, and clinical evaluations. It’s essentially an “everything you must…

Janet Woodcock, Judy McMeekin
During the past year, the U.S. Food and Drug Administration’s (FDA) approach to foreign and domestic inspections for food and medical products has been both risk-based and deliberate. The Covid-19 pandemic required us to rework our business operations so that we could carry out our public health…

Edmund Andrews
Seems everybody has a horror story about health insurance: Kafkaesque debates with robotic agents about what is and isn’t covered. Huge bills from a doctor you didn’t know was “out of network.” Reimbursements that take months to process.
It’s no secret that healthcare in the United States is…

Graham Freeman
Here’s an unfortunate truth: The story of the Covid-19 pandemic is one of epic quality failures in almost every area imaginable. Although there have been some admirable successes, such as the food and beverage organizations that have ensured the continued safe delivery of food supplies to most…
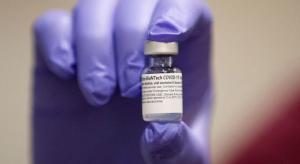
Rita Men
Ending the pandemic depends on achieving herd immunity, estimated at 70 percent or even 80 percent to 90 percent of a population. With some 30 percent of Americans telling pollsters they have no interest in getting vaccinated, that’s cutting it a bit close. The numbers are even worse in many other…