All Features

John O'Kelly
Warehouses are the backbone of supply chains, ensuring that goods move efficiently from suppliers to consumers. However, the physical demands of warehouse work—heavy lifting, repetitive motions, and prolonged standing—can take a toll on employees, leading to fatigue, injuries, and long-term strain…

Akli Adjaoute
Keeping quality high in today’s fast-moving production world is a big challenge. Traditional quality checks have worked well, but they can be slow, require a lot of work, and are prone to mistakes.
AI could change this by making quality control faster, more accurate, and easier to scale. It could…

ISO
How do health and safety incidents affect your business? If a worker is injured or becomes ill, what kind of disruption does it cause? Is your productivity affected? What’s the effect on other workers in terms of workload or psychological health and well-being?
People are the foundation of every…

Seb Murray
What happens when workplace colleagues land on opposing sides of an external competition? Henning Piezunka, an associate professor of management at Wharton, tackles this question in a recent study, using sports data to shed light on how outside rivalries can shape collaboration within the workplace…
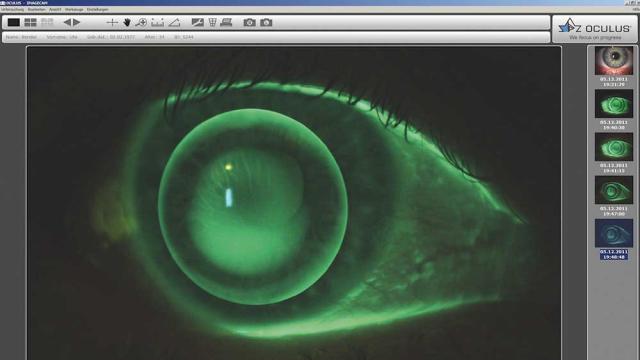
IDS Imaging Development Systems
The slit-lamp examination is one of the most important diagnostic techniques in ophthalmology. It enables a detailed examination of the anterior, middle, and posterior segments of the eye. Ophthalmologists can use it to recognize the smallest changes, anomalies, or damage. This procedure is used…
Matt McFarlane
The medical device industry is growing. Data from KPMG predict that global annual sales will rise by 5% per year to reach just under $800 billion by 2030. New technology, new opportunities, and, as always, the promise of improving patient outcomes around the world are major drivers of growth within…

John Tschohl
I ’ve been skiing in Vail, Colorado, one of the best skiing areas in the world, for more than 50 years. George Gillett Jr. acquired the resort in 1985. But when I first met him in 1978 and asked him what he does, he said, “I am in customer service.”
Gillett is out of that business now. And though…
Etienne Nichols
Have you ever wondered what your medtech company looks like from the point of view of a U.S. Food and Drug Administration investigator? Well, this is your chance to find out.
Greenlight Guru invited Vincent Cafiso, a former FDA investigator, to the Global Medical Device Podcast to share his…

Stephanie Ojeda
Quality risk management (QRM) has become a crucial tool for ensuring regulatory compliance worldwide. It plays a central role in ISO management system standards and regulations, as well as the EU Medical Device Regulation (MDR/IVDR), FDA 21 CFR 820, and ICH Q10 in the pharmaceutical and biotech…

Harry Hertz
My story begins with Hurricane Milton, one of two tragic hurricanes to hit the west coast of Florida last year. Milton went right over Sarasota, where I live part of the year. It was a devastating storm; tree and plant debris still remain on the sides of many roads. Big root balls are still upended…

Adam Grabowski
Cash is king for manufacturers, from the owner down to the machine operators. If you visit any manufacturer, you’ll see that most have a keen eye on how everything is being used. Machines are generally only running if they are making parts; employees are typically only working if orders are coming…

Siemens USA
Since the early 1980s, the automotive industry has used hydraulically actuated (servo-hydraulic) test systems to simulate operating speeds and road conditions for testing OEM components and fully assembled vehicles. These systems have helped unlock vast improvements in the quality, safety, and…

Wayne Labs
A September 2024 Gallup poll revealed that nearly 30% of U.S. adults have little to no confidence in the safety of U.S. foods at the grocery store. Considering recent recalls, is it any wonder? What good is a quality control program if it doesn’t include food safety?
Boar’s Head’s Jarratt,…

Sabine Terrasi
It creates a warm and inviting atmosphere. Its natural grain and color tones have a lively effect and add character to a room. We are talking about classic wood flooring—usually laminate, and optionally with a click system for easy installation.
Scheucher Holzindustrie GmbH, from Mettersdorf,…

Stephanie Ojeda
In April 2018, the U.S. Food and Drug Administration (FDA) approved the first artificial intelligence-powered diagnostic system, a software program used to detect diabetes-related vision loss.
Since then, the industry has seen explosive growth of AI in medical device manufacturing, which is…

Ilana J. Sprongl
Back in 2023, only 15% of businesses had adopted AI-augmented software testing tools. By 2027, that number is expected to leap to 80%. The reason behind this rapid adoption is clear. The complexity of modern software and products is skyrocketing, and with it, the risks associated with quality…

Akhilesh Gulati
In the world of operations and quality management, the pressure to act quickly can feel overwhelming. Senior executives are constantly racing against time to meet customer demands, solve problems, and keep shareholders satisfied. In the rush to address immediate challenges, “Ready, aim, fire!” gets…

4C Consulting
ISO 27001 is a globally recognized standard for establishing, implementing, maintaining, and improving an information security management system (ISMS). Successfully implementing ISO 27001 can provide tremendous benefits, such as ensuring data security, building trust with customers, and meeting…

Silas Fulsom
In today’s highly competitive global markets, lean manufacturing is no longer something that’s nice to have—it’s a way of life. You can either reduce costs, simplify operations, and improve productivity on a continual basis—or you can lose business to competitors that do.
Lean manufacturing starts…

Megan Wallin-Kerth
It’s 2024, and the age of automation has largely taken over our phones, computers, and businesses. This isn’t entirely a bad thing, you understand. It gives us the ability to spend time and money elsewhere—from investing in staff to spending more time on innovating technology rather than whittling…

William A. Levinson
The ongoing relevance of the quality profession requires evolution and adaptation to meet the needs of the 21st century. Remember, the quality profession originated with the need for inspection to prevent poor quality from reaching customers; this was before it evolved to include metrology,…

Etienne Nichols
Compliance with industry regulations and standards is a fundamental part of medtech. Without proper medical device compliance, companies risk patient harm, litigation, and reputational damage.
Fortunately, compliance with medical device regulations and standards is not an impossible task. A…

Mike Figliuolo
Everyone has goals these days. The one that causes more disconnects than any other is a sales goal. It’s a number to hit (either units or dollars or both—the better ones are actually measured in profit dollars rather than revenue dollars). They’re problematic because by their very nature they get a…
Victoria Alestra
In regulated industries like pharmaceuticals, medical devices, and food manufacturing, compliance is crucial for operational excellence. A validated quality management system (QMS) is key to maintaining this compliance. Let’s explore how QMS software streamlines validation and ensures regulatory…

Etienne Nichols
A quality management system (QMS) is at the heart of every successful medtech company. A QMS comprises all the policies, processes, and procedures that ensure the production of safe and effective medical devices—which means that problems with your QMS can quickly become problems with your products…