All Features

Cornelia C. Walther
On April 8, 2025, a driverless Zoox robotaxi misjudged an approaching vehicle, braked too late, and sideswiped it at 43 mph on the Las Vegas Strip.
One month later, the Amazon subsidiary issued a software recall on 270 autonomous vehicles and suspended operations while regulators investigated the…

Heidi Drafall
Anyone who has cracked their smartphone screen or had a rapid oil change knows that sometimes the OEM isn’t the most affordable or convenient service option. Consumer flexibility, paired with lower-cost, high-quality options, is logical, whether it’s in the consumer market or in healthcare.
The…
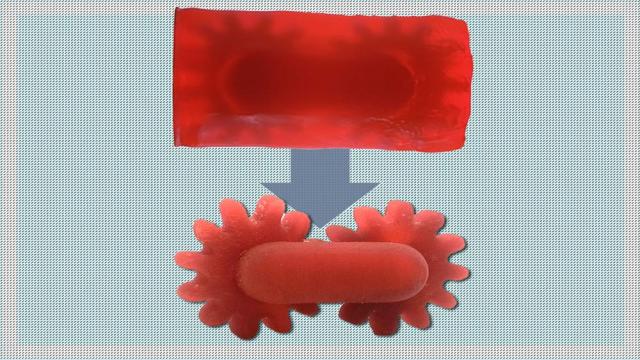
Jennifer Chu
Hearing aids, mouth guards, dental implants, and other highly tailored structures are often products of 3D printing. These structures are typically made via vat photopolymerization—a form of 3D printing that uses patterns of light to shape and solidify a resin, one layer at a time.
The process…

William A. Levinson
According to the U.S. News & World Report article “FDA Warns Sanofi of Manufacturing Irregularities at Key Facility” (Jan. 23, 2025), the pharmaceutical company Sanofi received a U.S. Food and Drug Administration warning letter “stating that FDA inspectors found irregularities with the facility…

Creaform
The Marshall Advanced Manufacturing Center (MAMC) is a leading-edge resource facility dedicated to driving innovation and advancing manufacturing technologies.
Operating from West Virginia facilities in Huntington, South Charleston, and Point Pleasant, the MAMC is at the heart of groundbreaking…

Kate Zabriskie
You know that friend who’s always there when you’re in need? The one who shows up on time, follows through on promises, and genuinely cares about what’s going on in your life? That’s exactly what your business needs to be for your customers.
Sure, those splashy marketing campaigns and point-…

Sunderesh Heragu
According to the U.S. Census Bureau and the U.S. Bureau of Economic Analysis, trade with our three largest partners—Canada, China, and Mexico—accounted for more than $1.32 trillion in imports and $0.82 trillion in exports in calendar year 2024. This represented 40% of the total trade between the…
Matt McFarlane
One of the key findings in Greenlight Guru’s 2025 Medical Device Industry Report was that economic uncertainty is playing a large role in the decisions medical device companies make this year.
The report surveyed more than 500 medical device professionals across quality, regulatory, product…

Jennifer Chu
For a robot, the real world is a lot to take in. Making sense of every data point in a scene can take a huge amount of computational effort and time. Using that information to then decide how to best help a human is an even thornier exercise.
Now, MIT roboticists have a way to cut through the data…

Bruce Hamilton
A few months ago I visited a potential customer, a high-tech startup, which like many Boston-area tech companies is developing astounding products that would have been considered science fiction only 10 years ago. The parking lot was half full at 8 a.m., but the entrance was locked to visitors, and…

Stephanie Ojeda
Every day, quality leaders face a variety of production and process issues. Although some problems are easy to fix, others require deeper investigation, such as using a 5 Whys analysis or fishbone diagram. But then there are the stubborn, recurring issues that can lead to quality issues, increased…

Akhilesh Gulati
When we step into a complex organization—whether in manufacturing, healthcare, or finance—we often find ourselves navigating a sea of competing truths. Everyone seems certain they see the problem clearly. Yet somehow, solving it feels harder than it should.
Why?
Often, it’s not the facts that…

ISO
Occupational health and safety (OHS) is often brushed aside as a checkbox exercise—something assigned to compliance officers or forgotten in day-to-day operations. But this mindset comes at a cost. Every year, millions of people suffer injuries, illnesses, or worse, simply because their workplace…

Troy Harrison
Salespeople are getting older. Buyers are getting younger. You have a disconnect.
In raw terms, the average age of a professional business-to-business (B2B) salesperson in the United States is 47.1 years old. Fifteen years ago that number was 42. So, the sales profession is graying. Meanwhile, the…

Mike Figliuolo
Nobody likes change. I know if you do anything that changes my routine in the morning, my whole day is whacked. We all hate change. Heck, most of us hate getting change at the grocery store because of all those coins.
As a leader, though, your job is to get others to want to change.
Getting other…

John O'Kelly
Warehouses are the backbone of supply chains, ensuring that goods move efficiently from suppliers to consumers. However, the physical demands of warehouse work—heavy lifting, repetitive motions, and prolonged standing—can take a toll on employees, leading to fatigue, injuries, and long-term strain…

Akli Adjaoute
Keeping quality high in today’s fast-moving production world is a big challenge. Traditional quality checks have worked well, but they can be slow, require a lot of work, and are prone to mistakes.
AI could change this by making quality control faster, more accurate, and easier to scale. It could…

ISO
How do health and safety incidents affect your business? If a worker is injured or becomes ill, what kind of disruption does it cause? Is your productivity affected? What’s the effect on other workers in terms of workload or psychological health and well-being?
People are the foundation of every…

Seb Murray
What happens when workplace colleagues land on opposing sides of an external competition? Henning Piezunka, an associate professor of management at Wharton, tackles this question in a recent study, using sports data to shed light on how outside rivalries can shape collaboration within the workplace…
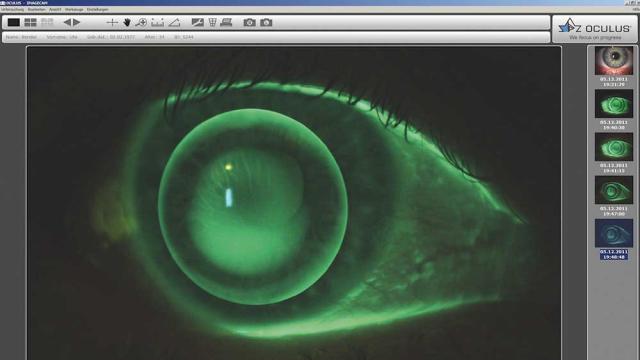
IDS Imaging Development Systems
The slit-lamp examination is one of the most important diagnostic techniques in ophthalmology. It enables a detailed examination of the anterior, middle, and posterior segments of the eye. Ophthalmologists can use it to recognize the smallest changes, anomalies, or damage. This procedure is used…
Matt McFarlane
The medical device industry is growing. Data from KPMG predict that global annual sales will rise by 5% per year to reach just under $800 billion by 2030. New technology, new opportunities, and, as always, the promise of improving patient outcomes around the world are major drivers of growth within…

John Tschohl
I ’ve been skiing in Vail, Colorado, one of the best skiing areas in the world, for more than 50 years. George Gillett Jr. acquired the resort in 1985. But when I first met him in 1978 and asked him what he does, he said, “I am in customer service.”
Gillett is out of that business now. And though…
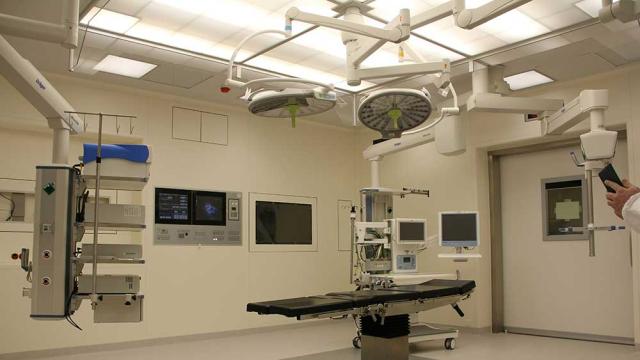
Etienne Nichols
Have you ever wondered what your medtech company looks like from the point of view of a U.S. Food and Drug Administration investigator? Well, this is your chance to find out.
Greenlight Guru invited Vincent Cafiso, a former FDA investigator, to the Global Medical Device Podcast to share his…

Stephanie Ojeda
Quality risk management (QRM) has become a crucial tool for ensuring regulatory compliance worldwide. It plays a central role in ISO management system standards and regulations, as well as the EU Medical Device Regulation (MDR/IVDR), FDA 21 CFR 820, and ICH Q10 in the pharmaceutical and biotech…

Harry Hertz
My story begins with Hurricane Milton, one of two tragic hurricanes to hit the west coast of Florida last year. Milton went right over Sarasota, where I live part of the year. It was a devastating storm; tree and plant debris still remain on the sides of many roads. Big root balls are still upended…