FOBA
With unique device identification (UDI) required under U.S. Food and Drug Administration regulations and the EU Medical Device Regulation, direct part marking has become mandatory for many medical devices, including surgical instruments and implants.
|
ADVERTISEMENT |
In practice, these requirements are challenged by highly reflective metals, extremely limited marking areas, and demanding conditions throughout the entire device life cycle. Markings must remain permanently high‑contrast and reliably legible without compromising function or material properties.
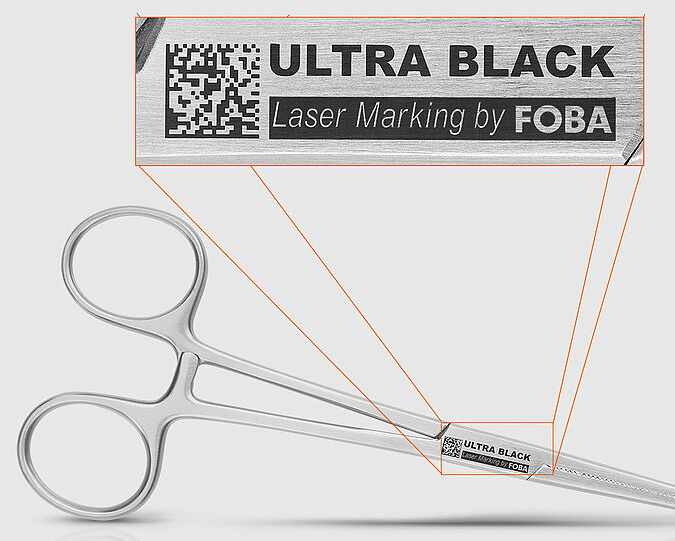
Ultrashort pulse laser black marking has emerged as a reliable solution to these challenges. The effect is used particularly on medical‑grade stainless steel, but it’s also suitable for other metals.
Four key challenges in direct part marking on medical‑grade stainless steel
Stainless steel is indispensable in medical technology due to its corrosion resistance, mechanical strength, and biocompatibility. And yet, direct part marking on medical‑grade stainless steel is technically demanding. Four aspects deserve particular attention.
1. Reflections on polished surfaces
High‑gloss finishes complicate direct marking, optical inspection, and code verification. Reflections reduce contrast and impair readability.
2. Extremely small marking fields and complex geometries
Micro‑instruments or functional surfaces often provide only minimal space. Codes must be exceptionally fine and precise, yet remain reliably readable.
3. Thermal effects and corrosion risk
Thermally induced marking effects can affect the passive layer and surface properties. The trade‑offs between contrast, corrosion resistance, and material integrity must be considered.
4. Exposure to reprocessing
Cleaning, disinfection, sterilization, and passivization repeatedly affect the surface. Markings must remain durable and corrosion‑free throughout the entire life cycle.
This combination of reflectivity, miniaturization, material sensitivity, and reprocessing stress means that conventional laser marking techniques (e.g., ablation or annealing) can reach their limits for certain marking requirements. This is exactly where black marking demonstrates its strengths, because it addresses all four challenges at once.
How black marking works: Nanostructures instead of heat input
Black marking refers to a laser marking effect that produces deep black, matte, nonreflective marks. A defining characteristic is angle‑ and illumination‑independent readability: The marking appears uniformly black regardless of viewing angle or lighting conditions. This is particularly relevant for vision‑based inspection processes and for reliable machine readability of DataMatrix codes commonly used for UDI marking.
“The black appearance is not created by material removal or a thermally generated oxide layer, but by a nanostructure on the surface. These so‑called ‘light traps’ reduce reflection, producing a strong contrast,” says Damian Zawadzki, product and application manager at FOBA Laser Marking + Engraving.
Black marking is typically performed using ultrashort pulse (USP) lasers. With ultrashort pulses in the femtosecond and picosecond range, and high pulse energy, the nanostructures required for the black marking effect form with virtually no heat input. Because the pulse duration is extremely short, very little energy is transferred into the surrounding material, and the process is often described as “cold” laser marking.
Long‑term durability can be demonstrated under realistic conditions. Extended testing conducted by medical technology service provider add’n solutions, together with FOBA Laser Marking + Engraving, did just that. Stainless steel instruments marked using the black‑marking process were repeatedly reprocessed (i.e., cleaning/passivation in a fully automated system, autoclaving, and additional highly alkaline cleaning intervals). The outcome: After 1,000 cycles, the markings created with the ultrashort‑pulse laser FOBA F.0100‑ir remained reliably legible.
“The marking is still excellently readable,” says Dominik Pfeiffer of add’n solutions. “It outlasts the service life of an instrument. In our test instruments, the material failed before the marking did.”
In summary, and in relation to the four challenges above, USP laser black marking combines reflection‑free contrast generation; high precision for miniaturized codes; extremely minimized heat input to protect the material; and strong resistance to cleaning, disinfection, and sterilization processes.
Black marking in practice: Application examples
Permanently legible UDI codes on high‑polish stainless steel instruments
Background: A surgical instrument manufacturer required a permanently durable, corrosion‑free UDI marking on a highly polished stainless steel surface. Conventional fiber‑laser methods delivered insufficient contrast or caused surface changes. Reflections also impaired machine readability. Even standard ultrashort‑pulse lasers didn’t meet the customer’s expectations.
Solution: The continuously adjustable pulse width of the ultrashort pulse laser FOBA F.0100‑ir enabled precise and optimal adaptation of the energy input to the material and surface condition. This produced a deep black, reflection‑free contrast without affecting material or function.
Added value: Durable, robust, corrosion‑free marking with reliably readable UDI codes—even after countless cleaning and sterilization cycles.
Reliable readability of miniaturized codes on dental implants
Background: A dental implant manufacturer needed to mark extremely small areas with high information density. Shiny surfaces complicated machine readability, and the mark had to be positioned precisely within the limited space.
Solution: The high precision of the ultrashort‑pulse laser FOBA F.0100‑ir and FOBA’s integrated vision system IMP (intelligent mark positioning), in combination with the MarkUS software, were decisive. With optimal parameter configuration, the laser generates fine black‑marking structures on minimal surface area. The nonreflective, deep black mark supports reliable machine readability. The software‑ and vision‑based FOBA workflow ensures precise automated positioning and code inspection.
Added value: Reliable readability even of the smallest codes, reduced scrap through accurate positioning, and a stable marking workflow.
Practical guidelines for process design and quality assurance in black marking
In regulated environments, marking quality alone isn’t enough. Equally important is that the overall marking workflow is stable and suitable for qualification. In practice, the following measures have proven effective for implementing black marking successfully.
Consider material and surface
Alloy composition, surface finish, and cleanliness influence the parameter range in which stable contrast can be achieved. Even minor changes in material or surface preparation can shift the operating window. Zawadzki says, “When running marking tests, always use parts in their real series‑production condition.”
Adapt parameters precisely to the material and application
A reliable black‑marking application requires careful adjustment of laser parameters such as pulse energy, pulse duration, repetition rate, and focal position. Testing on original parts is the most reliable way to obtain robust results. “Our laser experts in the application labs run multiple tests with different settings,” says Zawadzki. “This is how we determine the optimal parameters aligned with the customer’s requirements.”
Include downstream life cycle steps
The product life cycle (e.g., cleaning, sterilization, passivation) should be included in qualification from the outset to ensure marking safety over time.
Plan for inline inspection and documentation
Particularly for UDI applications, verification of code quality immediately after marking—via a laser-integrated vision system—is recommended. Vision‑based inline inspection reduces risks early, while software‑based process data strengthens audit readiness and traceability.
Treat marking, inspection, and documentation as one integrated system
Maximum safety and reliability result from a holistic approach throughout all steps, from part positioning to documentation. A closed‑loop marking workflow, such as FOBA’s workflow, reduces interfaces, simplifies validation, and increases stability. FOBA combines laser technology, software control, automated alignment, vision‑based inspection, and documentation into a coordinated end‑to‑end system.
Black marking at a glance: Technology and workflow
Ultrashort pulse laser black marking provides a technically compelling solution for the demanding direct part marking of metallic medical devices. Deep black, reflection‑free contrast, strong resistance to cleaning and reprocessing, and the cold marking procedure address core challenges in medical device identification. However, long‑term success depends not only on the marking effect itself, but also on a well‑engineered overall solution. Only a stable, qualification‑ready workflow ensures that markings continue to meet regulatory, functional, and safety‑critical requirements over time.

Add new comment