Ken Appel is the manager of regulated industries for Veriteq.
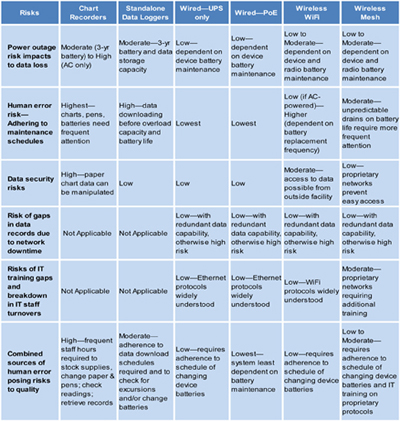
In part 1 of this article, we discussed the pros and cons of various systems for stand-alone monitoring instruments (e.g., chart recorders and data loggers) and wired networks, with and without power over Ethernet (POE). In the second part of this article, we will look at two types of wireless monitoring—Wi-Fi and mesh—and continue the discussion of the risk factors (figure 1) and costs of ownership (figure 2).
Click on image to enlarge.
Figure 1: General guidelines to risks associated with meeting good manufacturing practices (GMP) requirements
…
Add new comment