The U.S. Food and Drug Administration’s (FDA) Breakthrough Devices Program is beginning to show important results for patients since it was established in late 2016 under the 21st Century Cures Act to help patients gain timely access to breakthrough technologies.
|
ADVERTISEMENT |
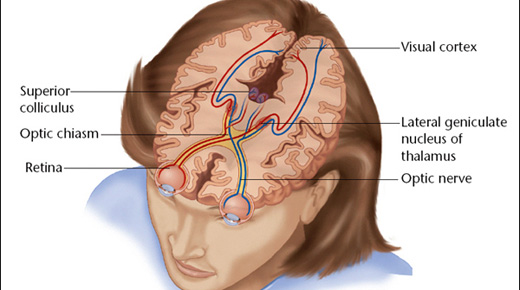
Consider Second Sight Medical Products Inc.’s Orion Cortical Visual Prosthesis System, which recently qualified for the FDA’s voluntary Breakthrough program.
Eligible technologies must provide more effective treatment or diagnosis for life-threatening or irreversibly debilitating diseases. In addition, there must be no approved or cleared treatment, or the device must offer significant advantages over existing approved or cleared alternatives.
…
Add new comment