
Quality by Design and RMM: Helping Manufacturers Balance Quality and Speed to MarketToday’s drug development companies face a difficult challenge: balancing the need for product quality and safety while speeding time to market. Championed by the FDA, EMEA, and other global regulatory agencies, quality by design (QbD) represents a systematic approach to building quality into product and process design and development right from the start. QbD and rapid-detection RMM go hand-in-hand, because both share the same goals--to ensure a high-quality manufacturing process that is lean, efficient, and reliable. Using RMM, manufacturers can more quickly test inbound raw materials. They are able to verify that any low levels of contamination are within the allowable threshold of microbial activity at the earliest stages, before a potentially compromised ingredient is combined with other materials. RMM saves downstream rework, and allows rapid testing of in-process and finished goods to serve as quality checkpoints along the way--validating that the product is coming together as designed. With rapid testing factored into the QbD equation, companies can significantly reduce their inventories and safety stock requirements, as well as dollars lost to costly product recalls or contaminated batches, and get quality products to market days faster. |
In today’s competitive marketplace, best-in-class manufacturers in the pharmaceutical, personal care products, consumer packaged goods, and beverage industries rely on lean manufacturing practices to achieve operational excellence. However, there remains a frequently overlooked opportunity.
By embracing next-generation rapid-detection microbiology testing methods (RMM), manufacturing companies can uncover measurable cost savings throughout the supply chain. At the same time, manufacturers can better meet or improve upon product quality standards and regulatory compliance policies.
How can this opportunity be realized and achieved? Before implementing RMM, companies need to understand the common challenges that can hinder production performance and increase costs.
For example, most manufacturers are conflicted by the need to be more responsive to their customers while improving overall profitability. They recognize that to increase customer satisfaction, they must reduce manufacturing lead times, shorten cycle times, decrease defects, and accelerate order fulfillment. Manufacturers must address internal priorities, such as reducing costs associated with excess inventory and safety stock, and strive to keep working-capital requirements to a bare minimum--in essence, use less to create more. This means cutting out excess waste.
The micro hold area: the last frontier for supply chain efficiency
Within manufacturing, one of the biggest culprits of waste is delays based on inefficient processes or movement of goods. Companies have invested millions of dollars on supply chain software and other costly initiatives. Yet they can literally cut days out of their cycle times by going to RMM, a process that quickly detects microbiological contamination in raw materials as well as in-process and finished goods.
Reducing waste and streamlining efficiencies require a closer look at the supply chain and, in particular, the micro department--the last frontier for process-time reduction. Makers of pharmaceuticals and personal care products, and other types of process manufacturers, put their inventories in quarantine for three, five, and even seven days. Many typically run micro tests using less expensive, slower agar plates to save costs, but the delays caused by lengthy micro hold times can cost a fortune in supply chain efficiency. Holding inventory for several days stymies customer responsiveness, creates waste on the production floor, and ties up valuable resources that could otherwise be contributing to profitability. When inventory isn’t getting off the floor and out the door fast enough, revenue is lost.
Consider how much companies spend to relocate processes on the manufacturing floor to save just 15 minutes in the production cycle. What would it be worth to save three days? That’s where the benefit of RMM becomes clear.
Next-generation RMM systems contain newer enzyme technology that significantly compresses testing times to 18-24 hours vs. three to seven days with traditional methods. Thanks to greater sensitivity and reproducibility, more advanced RMM technology offers a distinct advantage. By reducing the time required to test and release goods, manufacturing companies can drive new efficiencies throughout the supply chain and realize significant cost savings. Such technology also supports efforts to achieve certification with ISO 9001 standards, cGMP compliance, and other manufacturing best practices.
Next-generation RMM solutions come to light
Compared to traditional detection systems using agar plates (a process that has been used for more than 100 years), new RMM technology can greatly reduce manufacturing cycle times and enhance throughput. With agar plates, manufacturers wait for results from incubations of three to seven days in the micro hold area. Today’s RMM systems use adenosine triphosphate (ATP) and adenylate kinase (AK) bioluminescence assays that cut this waiting time by half or more. This significantly shortens cycle times.
Virtually anything that can be tested the traditional way can be tested with rapid methods; it’s just that some systems are more flexible than others. When evaluating rapid systems, look for one that can test the widest range of your materials--including filterable and nonfilterable samples. Ask if the system has been used to test similar products. Because most systems can readily detect the presence of a known contaminant, be sure to assess its value in absence screening, especially for slow-growing molds. Very few systems can ensure the absence of mold in 24 hours. Ideally, select a method where the test is nondestructive. This ensures that you can retest any sample that comes up positive.
For high-volume facilities, consider the system’s throughput. If samples must incubate in the testing unit for many hours, then the unit will be unavailable for testing a good portion of the time, and you may need to purchase additional units. Carefully examine the protocol itself. How simple and familiar will it be to your testing staff? Is it easy enough to be used by nonscientists? How much space does the system take up? If necessary, is the instrument robust enough to be run on the factory floor? Finally, does the company provide ongoing technical, scientific, and regulatory support worldwide?
ATP technology
Bioluminescence is best known from the glow of a firefly tail. It occurs when luciferase and luciferin come into contact with the ATP molecule, which is present in all living organisms. Rapid microbial screening systems use ATP bioluminescence and sensitive light instruments (luminometers) to reveal the presence of microbes (which contain ATP) in product samples. The result is an emission of light that is directly proportionate to the amount of ATP present in the sample. ATP bioluminescence technology has been around for 40 years, but only since the introduction of selective extraction reagents has the potential for this technology been tapped for use in a broad range of manufacturing industries. RMM results using ATP typically are available within 24-48 hours.
AK technology
The latest development in the field of bioluminescence is the use of the AK enzyme, which is another vital part of energy metabolism that is present in all living organisms. Because AK is an enzyme rather than a metabolite, it is possible to use AK to generate almost unlimited amounts of ATP, which can then be detected by a conventional bioluminescence assay. This new AK technology makes it possible to get test results in 18-24 hours, which is still a statistically significant improvement over current ATP-based screening methods that can take up to 48 hours.
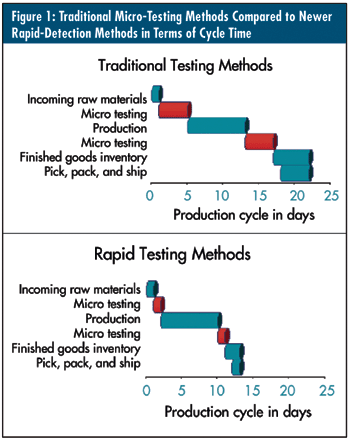
The diagrams in figure 1, above, compare traditional micro-testing methods to newer rapid-detection methods in the manufacturing production process. By compressing the time required for micro testing at various stages of production, manufacturers can significantly streamline cycle times. The cumulative effect of these cycle-time savings is substantial--up to 12 days in this case.
Containing contamination saves costs
Put simply, the results of microbial testing with new RMM technologies are now available to manufacturers in just hours--not days. This represents a new best practice for manufacturing companies whose livelihoods depend on cycle time speed and supply chain efficiency.
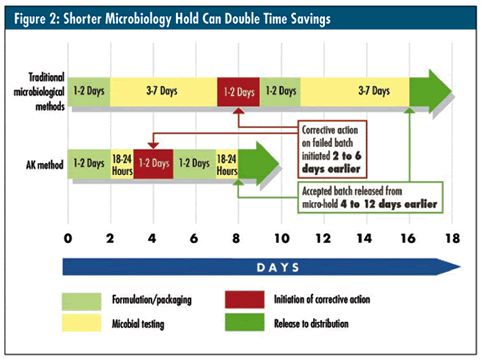
For example, earlier identification of contamination helps manufacturers to release quarantined stock or batch releases much sooner, as shown in figure 2, above. RMM also enables companies to bring new products to market faster, which presents a critical competitive benefit. Finally, the sooner a contamination event is identified, the sooner it can be contained. Manufacturers using RMM can initiate faster corrective action, reduce production recovery time, and provide for more rapid release of replacement product. In the end, quicker response time minimizes the overall economic consequence of discarded or reprocessed goods--saving companies not only money, but also reputation.
Create measurable value through RMM
Today’s rapid-detection methods provide manufacturers with quantifiable value.
Dramatic decreases in total microbiological testing time drives reduced working capital requirements, a streamlined supply chain, improved manufacturing efficiencies, and faster recovery from contamination events. Such improvements translate to cost savings that are measurable.
Following are some examples of operational areas where savings can be quantified as a result of implementing RMM:
• Reduces working capital requirements. RMM can help lower the cost of capital by decreasing your investment in quarantined inventory as well as safety stock due to shorter cycle times.
• Decreases stock carrying costs. What’s your interest rate for finished-goods capital? RMM helps reduce the amount of capital tied up in finished-goods inventory, and therefore shaves costs for carrying excess stock.
• Saves warehouse space required for quarantined inventory/safety stock. How much floor space is required to store one day’s production of finished-goods? To store inventory and safety stock? Calculate the cost of one unit of area of warehouse space per year and see how much can be saved through faster product release.
• Streamlines the supply chain. RMM drives finished goods to market faster, leading to quicker sales. This helps accelerate your revenue cycle.
• Improved recovery from contamination events. Rapid detection not only accelerates recovery time, but also helps reduce the percentage of scrapped product.
The model in figure 3 below reflects the reduced investment in quarantined inventory and more effective management of contamination events. As you can see, there’s an ongoing positive trend in savings.
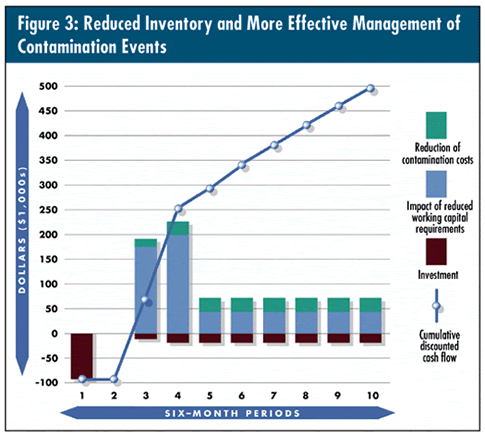
To quantify the value of implementing rapid methods, a global consumer products company, in conjunction with Arthur D. Little management consultants and Celsis Inc., developed a financial model that estimates the payback period and five-year net present value using the company’s specific manufacturing and testing data. Although results will vary based on company specifics--such as cost of capital and average daily output of finished goods--the typical single-plant operation will achieve payback in six-to-nine months and realize a net present value of $500,000-$750,000.
In conclusion
Faced with ever-increasing pressures to drive costs out of product fulfillment while meeting customer demands, best-in-class pharmaceutical, personal care products, and consumer packaged goods manufacturers can deploy next-generation RMM solutions that speed product-to-market time while ensuring that product quality and safety standards are met. Companies that incorporate value-added RMM technology into their micro departments will benefit from manufacturing and supply chain efficiencies. These translate to direct and measurable cost savings as well as a competitive advantage in today’s global marketplace.
Comments
Add new comment