Given the Food and Drug Administration’s (FDA) increased findings for companies that must comply to CFR 21 Part 820—“Quality system,” it’s curious that the oversight body has not offered much guidance about product design control, particularly concerning sections 820.30 and 820.40 of the regulation. Even in the FDA’s expanded document, “Design Control Guidance for Medical Device Manufacturers,” there’s no mention of controlling the device life cycle.
|
ADVERTISEMENT |
An important distinction and critical element of any design control that defines a product life cycle (PLC) is that it can play a critical role during a medical device audit or for demonstrating design-control compliance. Developing a PLC in tandem with a solid product development process (PDP) provides the necessary cornerstones for complete design-control and device-management compliance.
Following is a summary of the the basics of a PLC. Although each manufacturing and development environment may need to adjust the model to meet its specific needs, keep in mind that merging the PLC and PDP so that they align and complement each other will help establish a solid design-control platform.
General cradle-to-grave product life cycle
A basic product life cycle includes research, development, production, and end of life. These can certainly be expanded as needed to address a specific device design or manufacturing procedure, as well as device class, category, and classification. There can be subphases during product development, such as prototype, engineering, build, alpha, beta, and archive, but the main concern is to define the life cycle that best meets the overall objective as it tracks various products and versions of products through development, the marketplace, and archival after they are discontinued (i.e., the company must still support those products in the marketplace even though their selling cycles have ended).
The diagram in figure 1 provides the basic PLC model and a generalized flow of the cycle. Much of the life cycle flow should be overlaid with a device-development process, so although the PLC is discussed here, remember that both the PLC and PDP for a given manufacturing environment should be developed together so that the two will complement each other and support the company’s overall strategy for design control of devices.
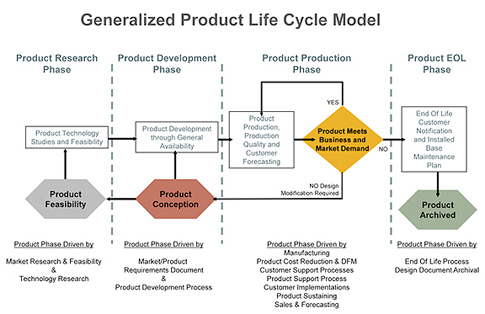
Figure 1: A basic product life cycle. (Source: Expandable Software) Click here for larger image.
The FDA allows some flexibility in design control as to when those controls should be implemented—usually somewhere in the gray area between research and development. This flexibility is mainly an attempt to loosen up compliance to encourage research into new technologies and devices for the medical industry. However, even during the research phase, a product manager needs some guidance to help define when a new technology should be developed for the marketplace, and when doing so would be most viable for the business.
For the rest of the PLC, the FDA will certainly want to see solid design controls and tracking of the device history, beginning with development and continuing through to the product’s end of life (EOL). Here, too, the PLC might include more specific EOL processes, depending on the devices and how they live beyond their final selling cycle.
Key points to consider when developing a specific PLC are:
1. It should be general enough to apply to finished product as well as supply-chain components.
2. Make sure the definitions for the research and development phases meet the required controls for a particular product as defined by the FDA. Note: the FDA can be a bit more flexible with the research phase as far as hard-and-fast design documentation, but make sure there is a clear distinction and definition built into a PLC and PDP and ensure adequate regulatory coverage.
3. Perform a clear and clean hand-off between the development and production departments and include all associated documentation. Be sure there is a process built into the PLC to review current products and verify that they meet both marketing and business needs. Allow for adjustments or changes as the product traverses its life cycle.
4. Design a process around its EOL, both from a business perspective (so as not to strand any inventory or leave customers without solutions) but also from a regulatory perspective, which emphasizes that no product really “ends”; rather, it gets archived and stored so that design history is retrievable.
From a product manager’s perspective, the earlier in a device’s manufacture and life that these principles can be applied, the better the product can be managed, from both a compliance and business perspective. Regardless of the FDA compliance, managing the product from a business perspective can pay big dividends in determining ideal time to market.
A short article can never do justice to defining a full PLC, but the key point here is that there is not a whole lot of FDA regulation or guidance for design control specific to PLC. A well-defined PLC can play a huge role in complying to medical device design control, especially when it’s overlaid with a well-defined PDP. When used in concert, the two processes can help companies avoid design control issues, FDA findings, or even warning letters or other punitive actions.
These PLC and PDP topics, the FDA’s perspective on design control normalization, and enterprise tools that can manage the process and can accelerate time to market are discussed in the white paper, “Quality System for Startup Medtech Companies: Design & Documentation Controls.”
First published Sept. 10, 2013, on the AssurX blog.
Add new comment